Search
Search our content by date or relevance.

Preparing for unannounced audits of medical devices
Published: 8 Jun 2023
ISO 13485 internal auditor Karandeep Singh Badwal, explains what to expect and how to prepare for unannounced audits of companies with a UKCA or CE-marked medical device.
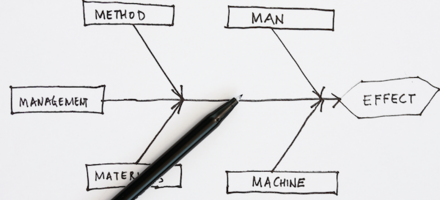
Quality tool: Cause and effect diagram
Published: 3 Feb 2021
Richard Green, CQP FCQI, Director of Kingsford Consultancy Services, UK, takes us through the Cause and Effect Diagram and how to apply the tool correctly.

Improving product reliability in manufacturing
Published: 28 Apr 2020
Dr Louis Redding, CQP FCQI, discusses some useful methods to help improve complex engineering products.

Quality World magazine
Published: 8 Aug 2016
The leading independent magazine dedicated to quality. Find out how to subscribe, read a sample below, or access this month's issue online now if you're a member of the CQI or IRCA.