Search
Search our content by date or relevance.

How to write clear and effective requirements
Principal auditor and Security Assurance Manager Dale Rollinson outlines the need for writing clear and effective requirements.

Quality management systems offering sustainable solutions to reduce waste
Murugesh Siva examines how quality management systems can assist in the key areas of managing both industrial and metal waste.

Adding value to audits of management systems
Senior auditor Pedro Mejias outlines five ways in which the audit process can create value to the auditee.

AI automation driving digital acceleration in testing
Mav Turner, CTO of DevOps at Tricentis, takes a closer look at the use of AI in quality assurance to drive software development.

Preparing for unannounced audits of medical devices
ISO 13485 internal auditor Karandeep Singh Badwal, explains what to expect and how to prepare for unannounced audits of companies with a UKCA or CE-marked medical device.

The importance of gap assessments in auditing
Conducting a gap assessment is a vital first step when implementing a new management system or integrating one with an existing system. IRCA Lead Auditor Andy Lau explains the steps that must be taken to ensure conformance with certification body standards.

Selecting the right problem-solving strategy
Derek Scott, CQP FCQI, Quality Manager at Rubberatkins, Scotland, takes a look at models and strategies for problem-solving.
Essentials for a safe on-site audit
Sharjeel Farooq, IRCA Principal Auditor, and Director of Advanced Certifications Pvt Limited in Pakistan, shares his experience and advice on how to hold a safe on-site audit during the Covid-19 pandemic.
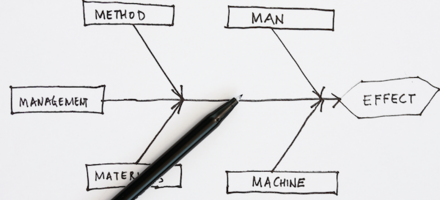
Quality tool: Cause and effect diagram
Richard Green, CQP FCQI, Director of Kingsford Consultancy Services, UK, takes us through the Cause and Effect Diagram and how to apply the tool correctly.

Managing quality and risk in projects
Abdullah Shaiban, Project Inspection Supervisor at Saudi Aramco – a multinational petroleum and natural gas company – highlights the importance of integrating risk into a quality management system.